GRANDER Research Concept
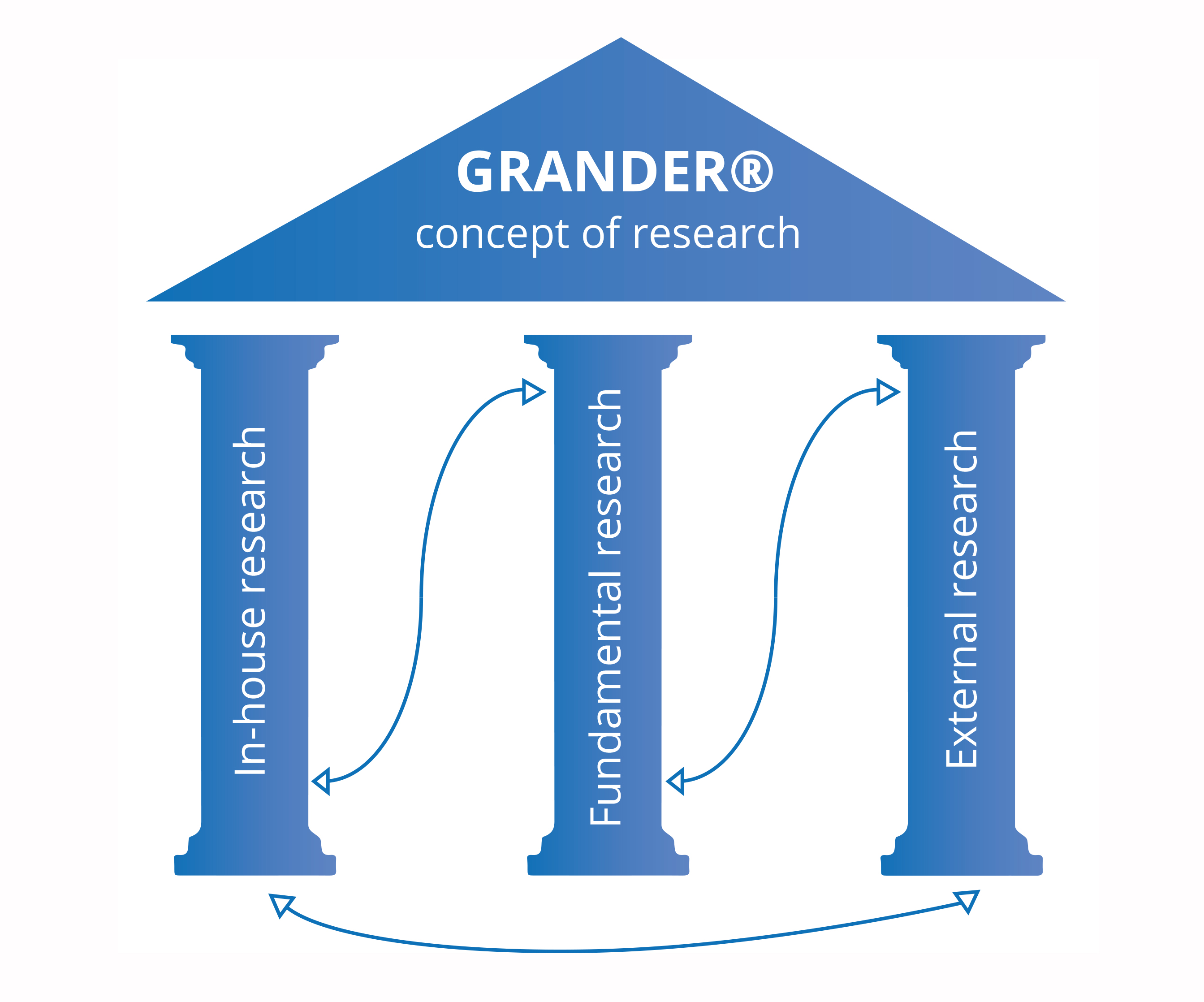 GRANDER lives up to the zeitgeist of approaching the world scientifically. Its new research concept has been named ‘Watertight’ – because it is exactly that. The program is built on three pillars and seeks to take great leaps in scientific discovery going forward. The program has a strong focus on scientific research. Surveys of subjective views are used only to a very small extent and are subject to strict scientific scrutiny.
GRANDER lives up to the zeitgeist of approaching the world scientifically. Its new research concept has been named ‘Watertight’ – because it is exactly that. The program is built on three pillars and seeks to take great leaps in scientific discovery going forward. The program has a strong focus on scientific research. Surveys of subjective views are used only to a very small extent and are subject to strict scientific scrutiny.
